Copper is a biologically essential trace element, which functions at the active sites of enzymes involved in electron transfer, processing of dioxygen, and in manipulation of certain nitrogen oxides. My group’s interest has focused on chemistry related to the latter two functions. Reversible copper(I)/dioxygen interactions occur in hemocyanin proteins, which are blood dioxygen carriers in molluscs and arthropods. Monooxygenase enzymes “activate” O2, promoting oxygen atom incorporation into biological substrates. Study of these enzymes or models for their action are also of interest in developing practical reagents or catalysts for oxidative transformations. Oxidases couple the reduction of O2 (to either hydrogen peroxide or water), while effecting one or two-electron substrate (e.g., alchohols, amines, other) oxidations. Copper ions also react with reduced dioxygen species such as superoxide (O2-), in Cu-Zn superoxide dismutases, and may also be involved in copper-mediate oxdiative damage in biological media, including possibly in Alzheimer’s disease.
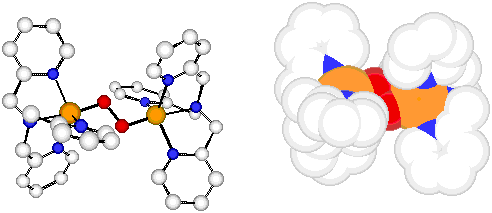
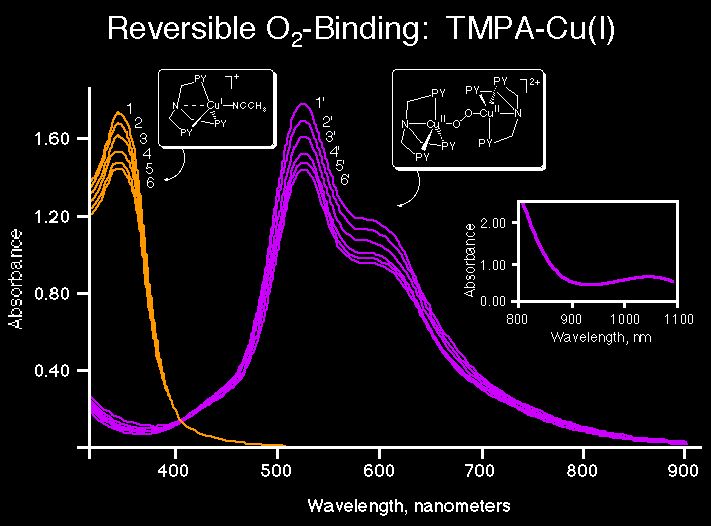
A primary focus in recent years has been the study of reversible dioxygen binding with copper complexes, to elucidate possible structures of Cu-O2species, their associated spectroscopy (UV-Vis, resonance Raman, etc.), and subsequent reactivity. To illustrate both the type of approach we use in our research, and some of the results obtained, it is instructive to provide some details of our work on a compound which provided the first x-ray crystallographic description for a copper-dioxygen adduct, synthetic or protein. [{(TMPA)CuII}2(O2-)]2+ (1), a Cu/O2 2:1 adduct, with PF6- or ClO4- counter-anions, forms by reversible oxygenation of the copper(I) precursor complex [(TMPA)CuI(RCN)]+ (1a, R = Me, Et, TMPA = tris[2-pyridylmethyl]amine). Product (1) is only stable at -80 C in solution or as a solid.
Physical Properties of {[(TMPA)CuII}2(O22-)]2+ (1)
X-ray: Cu…Cu = 4.36 ? Cu-O = 1.85 ? Angle: Cu-O-O = 107.7 ? O…O = 1.43 ?
Raman: nO-O = 834 cm-1 (18O2: 789 cm-1)
UV-Vis: lmax = 525 nm ( e = 11,500; p*s LMCT (O: Cu(II))
590 (sh) nm (e = 7,600) p*v LMCT (O: Cu(II))
1035 nm (e = 60) d-d band
Magnetism: EPR silent; Normal?1H NMR; Diamagnetic (-2J * 700 cm-1)
X-ray: J. Am. Chem Soc., 1988 110, 3690; J. Am. Chem. Soc., 1993, 115, 2677
Raman: J. Am. Chem. Soc., 1991, 113, 8671.
We have shown that the binding of O2 is reversible, and this can be illustrated by the spectroscopic experiments shown, wherein we can remove the bound O2 by application of a vacuum, and cycle back and forth between unoxygenated copper(I) complex (1a; lmax = 340 nm and (1; lmax = 525 nm, 600 nm) .
Some other completed or ongoing projects are:
1) Kinetic/thermodynamic investigations of dioxygen binding to copper(I) complexes, carried out in collaboration with the group of A. D. Zuberb�hler, Basel, Switzerland. Analogues of TMPA have been studied, including binucleating versions which hold two metal ion. These studies continue to provide important new insights, such as the characterization of intermediates LCu-O2, and the destabilization or enhanced stabilization of Cu2-O2 adducts afforded by particular ligands. The nature of the ligand is critical in determining the chemistry, just as the surroundings of a metal ion in a protein matrix (the enzyme) controls the chemistry.
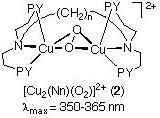
2) Tridentate ligands with nitrogen donors (PY = 2-pyridyl) afford side-on bridged peroxo-dicopper(II) complexes. Based on our study of model systems, we first proposed this O2-binding mode, and it has since been confirmed as the structure which occurs in hemocyanins.
3) Recent studies indicate that the side on peroxodicopper(II) complexes can be in equilibrium with bis-mu-oxo-dicopper(III) Cu2-(O)2 species. The factors which allow this interconversion are being actively pursued.
4) A major interest is the reactivity of Cu2-O2 or Cu2-(O)2 complexes. We have shown that the reactivity of end-on (as in 1) and side-on peroxo (i.e., (2)) complexes is very different. The former peroxo species is basic or nucleophilic in its reactions, while the latter is non-basic and electrophilic. We have previously extensively studied the mechanism of a model monooxygenase system, wherein a peroxo species like that in (2) attacks and hydroxylates an unactivated arene which is part of the ligand. More recently, we have discovered novel oxidative N-dealkylation chemistry (which is biomimetic) and are carrying out mechanistic studies. An exciting recent finding is that certain of our complexes can effect oxidations of externally added substrates, an important development in the pursuit of systems with practical applications.
5) Certain copper oxidases possess a trinuclear copper center, which functions to bind and reduce dioxygen. To understand the nature of these reactions, and for the possible development of reagents for oxidation of organics, ligands which bind three copper ions in close proximity are being synthesized and their copper chemistry developed.