Organic and polymer-based electronic materials hold great promise due to their wide range of electronic and other physical properties and the ability to control and tune them through functional modifications. We are working in collaboration with researchers in the JHU Departments of Materials Science and Engineering and Chemistry to combine synthesis, electronic characterization, and structural studies using x-ray and neutron diffraction to develop new classes of organic composite materials with novel functionality, and to control interfacial and electronic properties of thin films and semiconductor heterostructures. Current projects include development of small molecule/polymer composites with enhanced charge storage capabilities that have potential applications in areas ranging from piezoelectrics for energy harvesting to non-volatile organic field-effect transistors for memory, sensing, and logic elements.
- “Effect of organic electroactive crystallites in a dielectric matrix on the electrical properties of a polymer dielectric,” T. Lee, Y. Luo, C. Wang, A. Park, E. Zapata-Mercado, K. Hristova, T. Mueller, W. L. Wilson, D. H. Reich, and H. E. Katz, Phys. Rev. Materials 7, 065003 (2023).
- “Maximized hole trapping in a polystyrene transistor dielectric from a highly branched iminobis(aminoarene) side chain,” Q. Y. Zhang, B. Barrett, T. Lee, T. Mukhopadhyaya, C. C. F. Lu, E. C. Plunkett, T. Kale, C. Chi, K. J. T. Livi, P. McGuiggan, D. H. Reich, S. Thon, A. E. Bragg, and H. E. Katz, ACS Appl. Mater. Interfaces 13, 34584-34596 (2021).
- “Charge trapping in polymer electrets with highly dilute blended arylamine donors,”
E. C. Plunkett, Q. Y. Zhang, H. E. Katz, and D. H. Reich, ” ACS Appl Electronic Mater. 3, 1656-1662 (2021). - Effects of trifluoromethyl substituents on interfacial and bulk polarization of polystyrene gate dielectrics, E. Plunkett, T. S. Kale, Q Zhang, H. E. Katz, and D. H. Reich, Appl. Phys. Lett. 114, 023301 (2019).
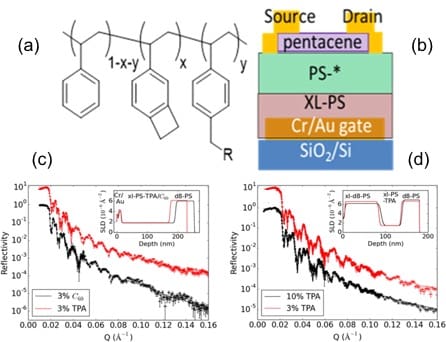
- Highly contrasting static charging and bias stress effects in pentacene transistors with polystyrene heterostructures incorporating oxidizable N,N’-bis(4-methoxyphenyl)aniline side chains as gate dielectrics, Q. Zhang, T. S. Kale, E. Plunkett, W. Shi, B. J. Kirby, D. H. Reich, and H. E. Katz, Macromolecules 51, 6011-6020 (2018).
- Synthesis, fabrication, and heterostructure of charged, substituted polystyrene multilayer dielectrics and their effects in pentacene transistors, O. J. Alley, E. Plunkett, T. S. Kale, X. Guo, G. McClintock, M. Bhuparthiraju, B. J. Kirby, D. H. Reich, and H. E. Katz, Macromolecules 49, 3478-3489 (2016).
