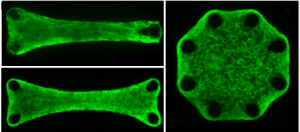
Systems that enable detailed experiments probing cellular response to and interactions with their 3D microenvironment are increasingly important in advancing studies of cellular mechanotransduction and tissue biomechanics. Our group has led the introduction and development of a novel bio-magnetomechanical system for magnetic actuation of arrays of 3D microtissues using microcantilevers. This system enables both in situ measurements of fundamental mechanical properties of engineered tissues, such as contractility and stiffness, as well as the response of cells to dynamic stimulation and matrix organization. Using this system, we have decoupled cell and extracellular matrix contributions to the tissue mechanical properties under both static and dynamic loading conditions, and studied the role of tissue geometry in determining matrix organization. Current projects include studies of cardiac and other cell types derived from human induced pluripotent stem cells, with applications to genetic-based cardiovascular disease.
- “Extracellular matrix alignment directs provisional matrix assembly and three dimensional fibrous tissue closure,” S. L. Das, P. Bose, E. Lejeune, D. H. Reich, C. Chen, and J. Eyckmans, Tissue Eng. Part A 27, 1447-1457 (2021).
- Effects of geometry on the mechanics and alignment of three-dimensional engineered microtissues, P. Bose, J. Eyckmans, T. D. Nguyen, C. S. Chen, and D. H. Reich, ACS Biomaterials Science & Engineering 5, 3843-3855 (2019).
- Matrix viscoplasticity and its shielding by active mechanics in a microtissue model: in situ experiments and mathematical modeling, A. S. Liu, H. Wang, C. R. Copeland, C. S. Chen, V. B. Shenoy, and D. H. Reich, Reports6, 33919(01-10) (2016).
- A microfabricated magnetic actuation device for mechanical conditioning of arrays of 3D microtissues, Xu, R. Zhao, A. S. Liu, T. Metz, Y. Shi, P. Bose, and D. H. Reich, Lab Chip 15, 2496-2505 (2015).
- Force-driven evolution of mesoscale structure in engineered 3D microtissues and the modulation of tissue stiffening, Zhao, C. S. Chen, and D. H. Reich, Biomaterials 35, 5056-5064 (2014).
- Decoupling cell and matrix mechanics in engineered microtissues using magnetically activated microcantilevers, R. Zhao, T. Boudou, W. G. Wang, C. S. Chen, and D. H. Reich, Advanced Materials 25, 1699-1705 (2013).
